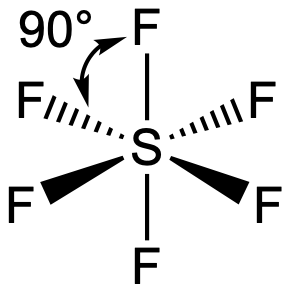
SF
6 contains 6 bonded and 0 nonbonded electron domains, giving octahedral e
- domain and molecular geometries. (AX
6E
0). The central atom lies at the center of the molecule. Four atoms can be visualized as lying in a common "square plane", forming 90 degree bond angles to the other atoms in the plane and the two atoms oriented perpendicular to the plane. All of the terminal atoms are in chemically equivalent positions, meaning there are no distinguishable equatorial and axial positions since the molecule is completely symmetrical. The molecule can be rotated in such a fashion that three unique combinations of four atoms can be visualized lying in a common plane (see check boxes below), with the other two atoms perpendicular to the plane. Each terminal atom in the molecule forms 90 degree bond angles with 4 other terminal atoms, and a 180 degree bond angle with the terminal atom directly across.
Use your mouse (computer) or fingers (touch screen) to manipulate the Jmol model.