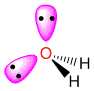
H
2O contains 2 bonded and 2 nonbonded electron domains, giving a tetrahedral e
- domain geometry and a bent molecular geometry. (AX
2E
2). The H-O-H bond angle (104 degrees) is less than in a perfect tetrahedron because the lone pair electrons spread out more in space than bonded electron pairs, compressing the bond angle. A cartoon model of the electron density of the lone pairs of electrons, represented by translucent purple spheroids, can be toggled on and off. Note that the shapes of the spheroids do not represent the actual electron densities of the lone pairs, but the shapes allow you to visualize how the presence of the lone pairs dictates the molecular geometry.
Use your mouse (computer) or fingers (touch screen) to manipulate the Jmol model.